Daughters of DES: A Cautionary Tale
A common sentiment we hear at the store is, “I was exposed to X when I was a kid and I'm fine” or “my mom did X when she was pregnant with me and I'm fine.” We sincerely hope that for everyone that says this, they really are “fine”. For the women who took the prescription drug DES during their pregnancies, mostly in the 1940s – 1960s, they thought they were fine as well. It was only after their daughters began to have alarmingly high rates of cervical and vaginal cancer decades later that they knew there was a problem.
As a mother of two daughters, I worry so much that something I have exposed them to could be harmful to them years from now or even to THEIR daughters. The best I can do is try to stay on top of the latest research, remember that not everything on the market is safe, and use Sprout to share what I learn with as many families as I can.
What is DES?
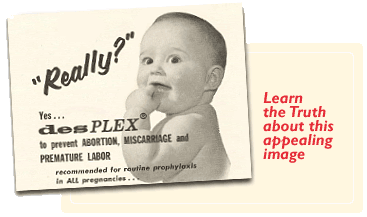
In 1938, DES (diethylstilbestrol) was the first synthetic estrogen to be created. It was promoted to doctors, who were urged to prescribe it to their pregnant patients. Never patented, DES was marketed using hundreds of brand names in the mistaken belief it prevented miscarriages and premature deliveries.
DES was prescribed primarily between 1938 and 1971 to prevent miscarriage, premature labor, and related complications of pregnancy. The use of DES declined after studies in the 1950s showed that it was not effective in preventing these problems.
In the early 1970s cases of a rare vaginal/cervical cancer were being diagnosed in young women. A cluster of them at Massachusetts General Hospital in Boston raised alarm among doctors who couldn't figure out what was going on. It took a persistent DES Mother to unravel the mystery. She told doctors her belief that the DES she was prescribed while pregnant was responsible. Ultimately, she was proven right.
In November 1971 the FDA told doctors to stop prescribing DES for their pregnant patients, however it was never banned. Specifically, the FDA said DES was contraindicated for pregnancy use. In some rare cases American doctors either didn’t hear of, or simply ignored the message and continued prescribing DES. Internationally, DES use during pregnancy continued for many subsequent years. In September 2000 the FDA withdrew approval of DES.
The known health effects for daughters born to mothers who took DES while pregnant are an increased risk for:
- ~ Clear cell adenocarcinoma (CCA), a rare kind of vaginal and cervical cancer
- ~ Reproductive tract structural differences (for example, T-shaped uterus)
- ~ Pregnancy complications, including ectopic (tubal) pregnancy and pre-term (early) delivery
- ~ Infertility
DES is now known to be an endocrine-disrupting chemical, one of a number of substances that interfere with the endocrine system to cause cancer, birth defects, and other developmental abnormalities. The effects of endocrine-disrupting chemicals are most severe when exposure occurs during fetal development. Increased risks of clear cell adenocarcinoma of the vagina and cervix have been found for daughters of women who took DES during pregnancy; fertility problems are also more common among these daughters. The daughters of women who used DES while pregnant—commonly called DES daughters—have about 40 times the risk of developing clear cell adenocarcinoma of the lower genital tract than unexposed women. Research has shown that the risk of developing this disease remains elevated as women age into their 40s.
Now researchers are investigating whether DES health issues are extending into the next generation, the so-called DES Grandchildren. As study results come in, there is growing evidence that this group has been adversely impacted by a drug prescribed to their grandmothers.
What could this mean for you?
DES daughters have an increased risk of developing abnormal cells in the cervix and the vagina that are precursors of cancer (dysplasia, cervical intraepithelial neoplasia, and squamous intraepithelial lesions). These abnormal cells resemble cancer cells, but they do not invade nearby healthy tissue and are not cancer. They may develop into cancer, however, if left untreated. Scientists estimated that DES-exposed daughters were 2.2 times more likely to have these abnormal cell changes in the cervix than unexposed women. It has been recommended that DES daughters have a yearly Pap test and pelvic exam to check for abnormal cells.
Several studies have found increased risks of premature birth, miscarriage, and ectopic pregnancy associated with DES exposure. Some studies suggest that the increased risk of infertility is mainly due to uterine or fallopian tube problems.
DES daughters should be aware of these health risks, share their medical history with their doctors, and get regular physical examinations.
To see if you could have been exposed, take a self assessment.
Sources:
http://www.cdc.gov/des/consumers/about/index.html
http://www.desaction.org/aboutdes.htm
http://www.cancer.gov/cancertopics/factsheet/Risk/DES